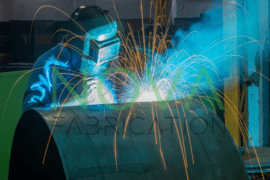Have you ever accidentally set your hand sanitizer on fire? No? Well, good for you. You’re probably using it right! But in any case you are wondering, yes, that cleaning liquid is actually fuel for the fire.

Science YouTuber Steve Mould shows that igniting hand sanitizer creates an almost invisible flame. Dim the lights and set your camera to proper settings and you can capture the outlines of a light blue flame flickering just above the sanitizer.
The culprit behind this unique flame is ethanol, a substance commonly found in cleaning agents such as ethyl alcohol. It can even be found in small quantities in a couple of alcoholic beverages and foods.

Normal red and orange flames usually have a lot of substances mixed with them. The chief substance which contributes to their color, Mould explains, is soot. Considering most readily flammable objects consist of different materials, igniting them causes the flames to burn impurely. Ethanol flame, on the other hand, is nearly invisible due to the fact that ethanol is burned into carbon dioxide which can’t be seen with the naked eye.

While it does give off a light blue flame, the heat emanating from burning ethanol isn’t hotter than any other visible fire. In fact, igniting your hand sanitizer results in a flame that is actually cooler than normal. It’s still hot and can burn you if you touch it though, so be careful when meddling with invisible flames!
This opens up a ton of possibilities for cool experiments. Say the world goes back to normal again; you can take a bottle of ethyl alcohol and use it to create a line of invisible flame to impress your friends at barbecues. You can even have fun solo and just indulge in lighting fireworks with the clear flame once the New Year comes around.
No matter what you decide to do with this newfound knowledge, just remember that this is the fire you’re playing with. It is smokeless and invisible, so completely extinguishing it can be hard. So please don’t go all caveman and burn everything in sight!